

GETTING STARTED: THE BASICS
Water, The chemistry of life.
Whenever we attempt to determine whether there is life as we know it on Mars or other planets, scientists first seek to establish whether or not water is present. Why? Because life on earth totally depends on water.
A High percentage of living things, both plant and animal are found in water. All life on earth is thought to have arisen from water. The bodies of all living organisms are composed largely of water. About 70 to 90 percent of all organic matter is water.
The chemical reactions in all plants and animals that support life take place in a water medium. Water not only provides the medium to make these life sustaining reactions possible, but water itself is often an important reactant or product of these reactions. In short, the chemistry of life is water chemistry.
Water, the universal solvent
 Water is a universal, superb solvent due to the marked polarity of the water molecule
and its tendency to form hydrogen bonds with other molecules. One water molecule,
expressed with the chemical symbol H2O, consists of 2 hydrogen atoms and 1 oxygen
atom.
Water is a universal, superb solvent due to the marked polarity of the water molecule
and its tendency to form hydrogen bonds with other molecules. One water molecule,
expressed with the chemical symbol H2O, consists of 2 hydrogen atoms and 1 oxygen
atom.
 Standing alone, the hydrogen atom contains one positive proton at its core with one
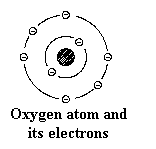
negative electron revolving around it in a three-dimensional shell. Oxygen, on the
other hand, contains 8 protons in its nucleus with 8 electrons revolving around it.
This is often shown in chemical notation as the letter O surrounded by eight dots
representing 4 sets of paired electrons.
Standing alone, the hydrogen atom contains one positive proton at its core with one
negative electron revolving around it in a three-dimensional shell. Oxygen, on the
other hand, contains 8 protons in its nucleus with 8 electrons revolving around it.
This is often shown in chemical notation as the letter O surrounded by eight dots
representing 4 sets of paired electrons.
The single hydrogen electron and the 8 electrons of oxygen are the key to the chemistry of life because this is where hydrogen and oxygen atoms combine to form a water molecule, or split to form ions.
Hydrogen tends to ionize by losing its single electron and form single H+ ions which are simply isolated protons since the hydrogen atom contains no neutrons. A hydrogen bond occurs when the electron of a single hydrogen atom is shared with another electronegative atom such as oxygen that lacks an electron.
Polarity of water molecules
In a water molecule, two hydrogen atoms are covalently bonded to the oxygen atom. But because the oxygen atom is larger than the hydrogens, its attraction for the hydrogen's electrons is correspondingly greater so the electrons are drawn closer into the shell of the larger oxygen atom and away from the hydrogen shells. This means that although the water molecule as a whole is stable, the greater mass of the oxygen nucleus tends to draw in all the electrons in the molecule including the shared hydrogen electrons giving the oxygen portion of the molecule a slight electronegative charge.
 The shells of the hydrogen atoms, because their electrons are closer to the oxygen,
take on a small electropositive charge. This means water molecules have a tendency
to form weak bonds with water molecules because the oxygen end of the molecule
is negative and the hydrogen ends are positive.
The shells of the hydrogen atoms, because their electrons are closer to the oxygen,
take on a small electropositive charge. This means water molecules have a tendency
to form weak bonds with water molecules because the oxygen end of the molecule
is negative and the hydrogen ends are positive.
A hydrogen atom, while remaining covalently bonded to the oxygen of its own molecule, can form a weak bond with the oxygen of another molecule. Similarly, the oxygen end of a molecule can form a weak attachment with the hydrogen ends of other molecules. Because water molecules have this polarity, water is a continuous chemical entity.
These weak bonds play a crucial role in stabilizing the shape of many of the large molecules found in living matter. Because these bonds are weak, they are readily broken and re-formed during normal physiological reactions. The disassembly and re-arrangement of such weak bonds is in essence the chemistry of life.
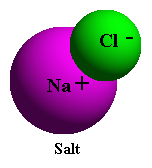 To illustrate water's ability to break down other substances, consider the simple
example of putting a small amount of table salt in a glass of tap water. With dry salt
(NaCl) the attraction between the electropositive sodium (Na+) and electronegative
chlorine (Cl-) atoms of salt is very strong until it is placed in water. After salt is
placed in water, the attraction of the electronegative oxygen of the water molecule
for the positively charged sodium ions, and the similar attraction of the electropositive
hydrogen ends of the water molecule for the negatively charged chloride ions, are
greater than the mutual attraction between the outnumbered Na+ and Cl- ions. In
water the ionic bonds of the sodium chloride molecule are broken easily because of
the competitive action of the numerous water molecules.
To illustrate water's ability to break down other substances, consider the simple
example of putting a small amount of table salt in a glass of tap water. With dry salt
(NaCl) the attraction between the electropositive sodium (Na+) and electronegative
chlorine (Cl-) atoms of salt is very strong until it is placed in water. After salt is
placed in water, the attraction of the electronegative oxygen of the water molecule
for the positively charged sodium ions, and the similar attraction of the electropositive
hydrogen ends of the water molecule for the negatively charged chloride ions, are
greater than the mutual attraction between the outnumbered Na+ and Cl- ions. In
water the ionic bonds of the sodium chloride molecule are broken easily because of
the competitive action of the numerous water molecules.
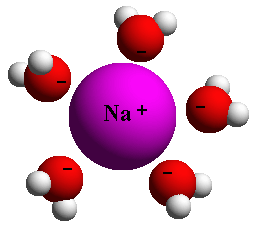
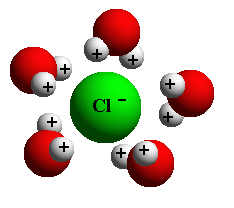
As we can see from this simple example, even the delicate configuration of individual water molecules enables them to break relatively stronger bonds by converging on them. This is why we call water the universal solvent. It is a natural solution that breaks the bonds of larger, more complex molecules. This is the chemistry of life on earth, in water and on land.
Oxidation-reduction reactions
Basically, reduction means the addition of an electron (e-), and its converse, oxidation means the removal of an electron. The addition of an electron, reduction, stores energy in the reduced compound. The removal of an electron, oxidation, liberates energy from the oxidized compound. Whenever one substance is reduced, another is oxidized.
To clarify these terms, consider any two molecules, A and B, for example.

When molecules A and B come into contact, here is what happens:
- B grabs an electron from molecule A.
- Molecule A has been oxidized because it has lost an electron.
- The net charge of B has been reduced because it has gained a negative electron (e-).
In biological systems, removal or addition of an electron constitutes the most frequent mechanism of oxidation-reduction reactions. These oxidation-reduction reactions are frequently called redox reactions.
Acids and Bases
An acid is a substance that increases the concentration of hydrogen ions (H+) in water. A base is a substance that decreases the concentration of hydrogen ions, in other words, increasing the concentration of hydroxide ions OH-.
The degree of acidity or alkalinity of a solution is measured in terms of a value known
as pH, which is the negative logarithm of the concentration of hydrogen ions:
What is pH?
On the pH scale, which ranges from 0 on the acidic end to 14 on the alkaline end, a solution is neutral if its pH is 7. At pH 7, water contains equal concentrations of H+ and OH- ions. Substances with a pH less than 7 are acidic because they contain a higher concentration of H+ ions. Substances with a pH higher than 7 are alkaline because they contain a higher concentration of OH- than H+. The pH scale is a log scale so a change of one pH unit means a tenfold change in the concentration of hydrogen ions.
Importance of balancing pH
Living things are extremely sensitive to pH and function best (with certain exceptions, such as certain portions of the digestive tract) when solutions are nearly neutral. Most interior living matter (excluding the cell nucleus) has a pH of about 6.8.
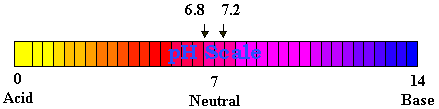
Blood plasma and other fluids that surround the cells in the body have a pH of 7.2 to 7.3. Numerous special mechanisms aid in stabilizing these fluids so that cells will not be subject to appreciable fluctuations in pH. Substances which serve as mechanisms to stabilize pH are called buffers. Buffers have the capacity to bond ions and remove them from solution whenever their concentration begins to rise. Conversely, buffers can release ions whenever their concentration begins to fall. Buffers thus help to minimize the fluctuations in pH. This is an important function because many biochemical reactions normally occurring in living organisms either release or use up ions.

Proceed to page 2.
Microwater Menu page
NOTE:
Dr. Hayashi is a Heart Specialist and Director of the Water Institute of Japan.
Dr. Hayashi has no affiliation with "Total Health Marketing",
or "Microwater Systems".